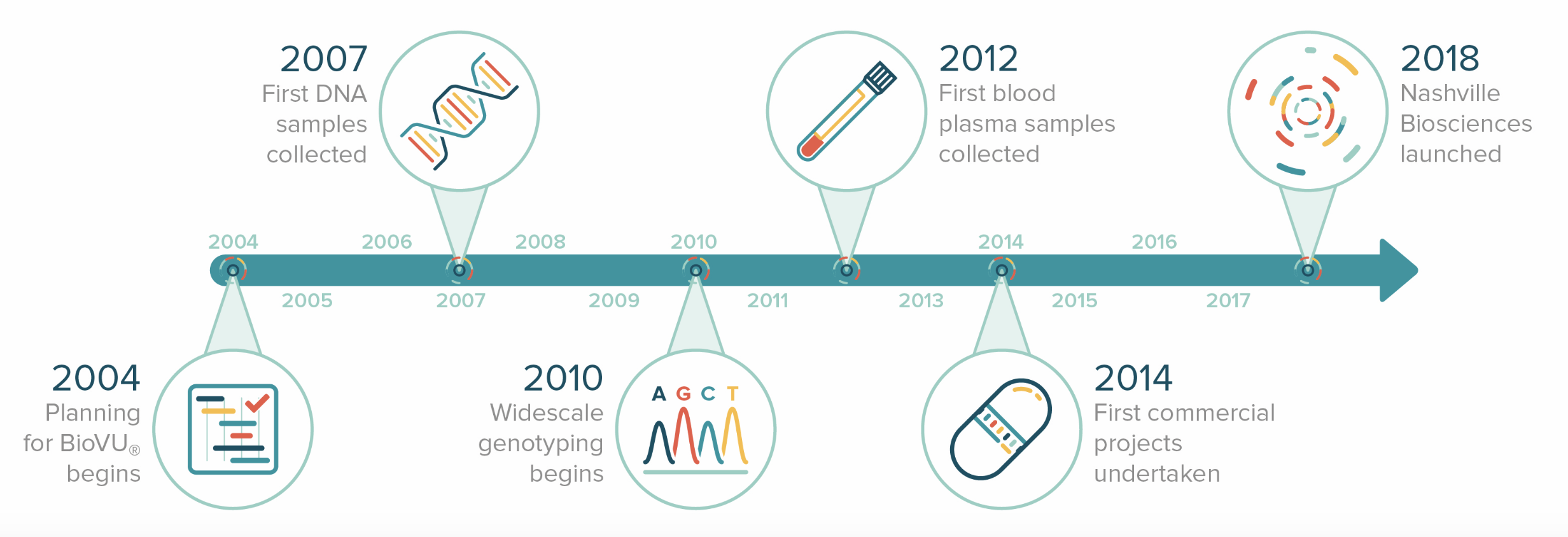
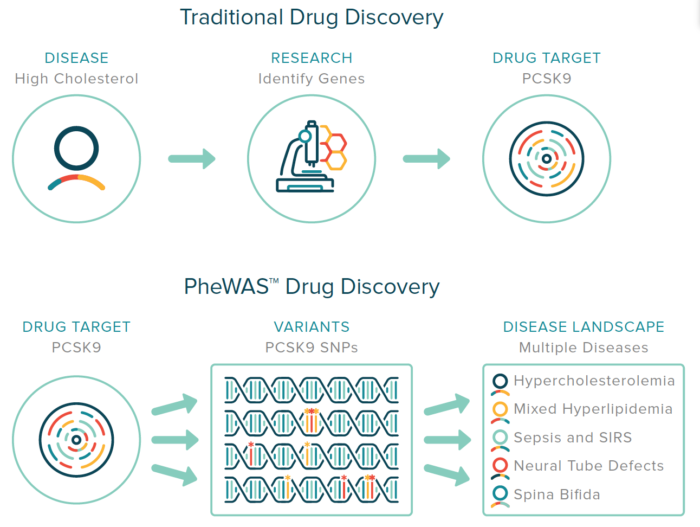
Patient welfare is a top priority for Nashville Biosciences and as such, we are committed to transparency in our practices and ensuring the protection of patient data.
Prior to any blood test, patients are provided with information about BioVU®, its research and goals. After learning about the program, patients are presented with a consent form in which they can either choose to donate their leftover blood sample or decline to participate. If a patient does not want to donate any leftover samples to BioVU®, they simply do not sign the form. If a patient consents to share his or her leftover samples with BioVU® and is a match for current research programs, BioVU® removes all personally identifiable information from the sample and medical record, then stores it in a safe and secure location. Additionally, if a patient changes their mind after signing the BioVU® consent form, they can call a BioVU® helpline to withdraw their participation from any future research.
Nashville Biosciences, Vanderbilt University Medical Center, and BioVU® regularly engage experts in ethics, science and research, operations, data security, and patient advocacy, among others, to ensure the accessibility and transparency of our information and to inform engagement with all stakeholders, including patients.